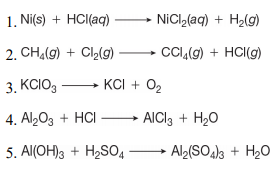
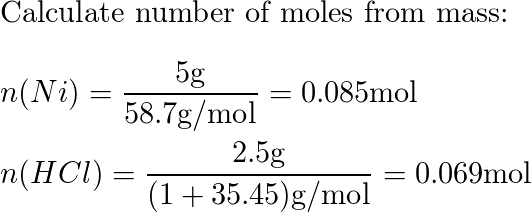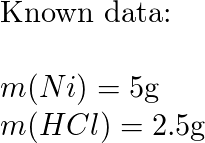a) Draw the mechanism for the reaction of tin/HCl with m-nitroacetophenone. b) Why is sodium hydroxide added to the reaction? What tin compounds are produced? How do you separate the tin salts

Answer in one sentence. Predict the product of the following reaction. Nitrobenzene→Sn/conc⋅HCl? - Chemistry | Shaalaa.com

Two-Electron HCl to H2 Photocycle Promoted by Ni(II) Polypyridyl Halide Complexes | Journal of the American Chemical Society

Effect of HCl concentration on desorption of Ni(II) from DHHC (amount... | Download Scientific Diagram

science chemistry precipitation reaction nickel hydrochloric acid | Fundamental Photographs - The Art of Science

a) Draw the mechanism for the reaction of tin/HCl with m-nitroacetophenone. b) Why is sodium hydroxide added to the reaction? What tin compounds are produced? How do you separate the tin salts
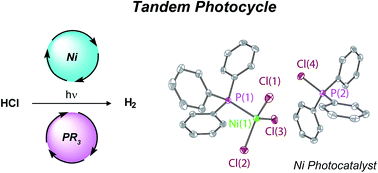
Tandem redox mediator/Ni(ii) trihalide complex photocycle for hydrogen evolution from HCl - Chemical Science (RSC Publishing)

Structure characterization of the Ni–C catalysts. (a) XRD patterns of... | Download Scientific Diagram
![A given nitrogen containing aromatic compound A reacts with \\[{\\text{Sn\/ HCl}}\\], followed by \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] to give an unstable compound B. B, on treatment with phenol, forms a beautiful coloured compound C with molecular A given nitrogen containing aromatic compound A reacts with \\[{\\text{Sn\/ HCl}}\\], followed by \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] to give an unstable compound B. B, on treatment with phenol, forms a beautiful coloured compound C with molecular](https://www.vedantu.com/question-sets/b565ff56-4b65-4f02-a304-a282cc9558db4662046607693484074.png)