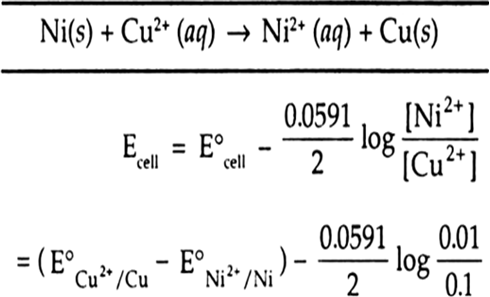What does NI(S) mean? - Definition of NI(S) - NI(S) stands for Naval Instructions to Salaried Consular Officers. By AcronymsAndSlang.com

A porous proton-relaying metal-organic framework material that accelerates electrochemical hydrogen evolution | Nature Communications

Engineering Sulfur Vacancies of Ni3S2 Nanosheets as a Binder-Free Cathode for an Aqueous Rechargeable Ni-Zn Battery | ACS Applied Energy Materials

Surface Activation and Ni‐S Stabilization in NiO/NiS2 for Efficient Oxygen Evolution Reaction - Zhang - 2022 - Angewandte Chemie International Edition - Wiley Online Library
XPS spectra of (Fe,Ni) 3 S 4 : (a) Fe 2p fresh, (b) Fe 2p calcined at... | Download Scientific Diagram

One-Step Synthesis of Nickel Sulfides and Their Electrocatalytic Activities for Hydrogen Evolution Reaction: A Case Study of Crystalline h-NiS and o-Ni9S8 Nanoparticles | ACS Applied Energy Materials

Calculate the `EMF` of the cell in whiCHM the following reaction takes place `:` `Ni(s)+2Ag^(o+)... - YouTube

The FTIR spectra of NH2-NI/S and NH2-NI/S after adsorption of Pb(II)... | Download Scientific Diagram

Heterogeneous histories of Ni‐bearing pyrrhotite and pentlandite grains in the CI chondrites Orgueil and Alais - Berger - 2016 - Meteoritics & Planetary Science - Wiley Online Library

Surface Restructuring of Nickel Sulfide Generates Optimally Coordinated Active Sites for Oxygen Reduction Catalysis - ScienceDirect
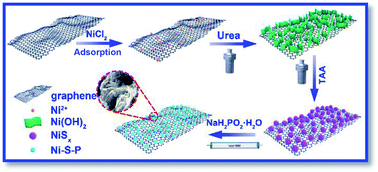
Anionic P-substitution toward ternary Ni–S–P nanoparticles immobilized graphene with ultrahigh rate and long cycle life for hybrid supercapacitors - Journal of Materials Chemistry A (RSC Publishing)


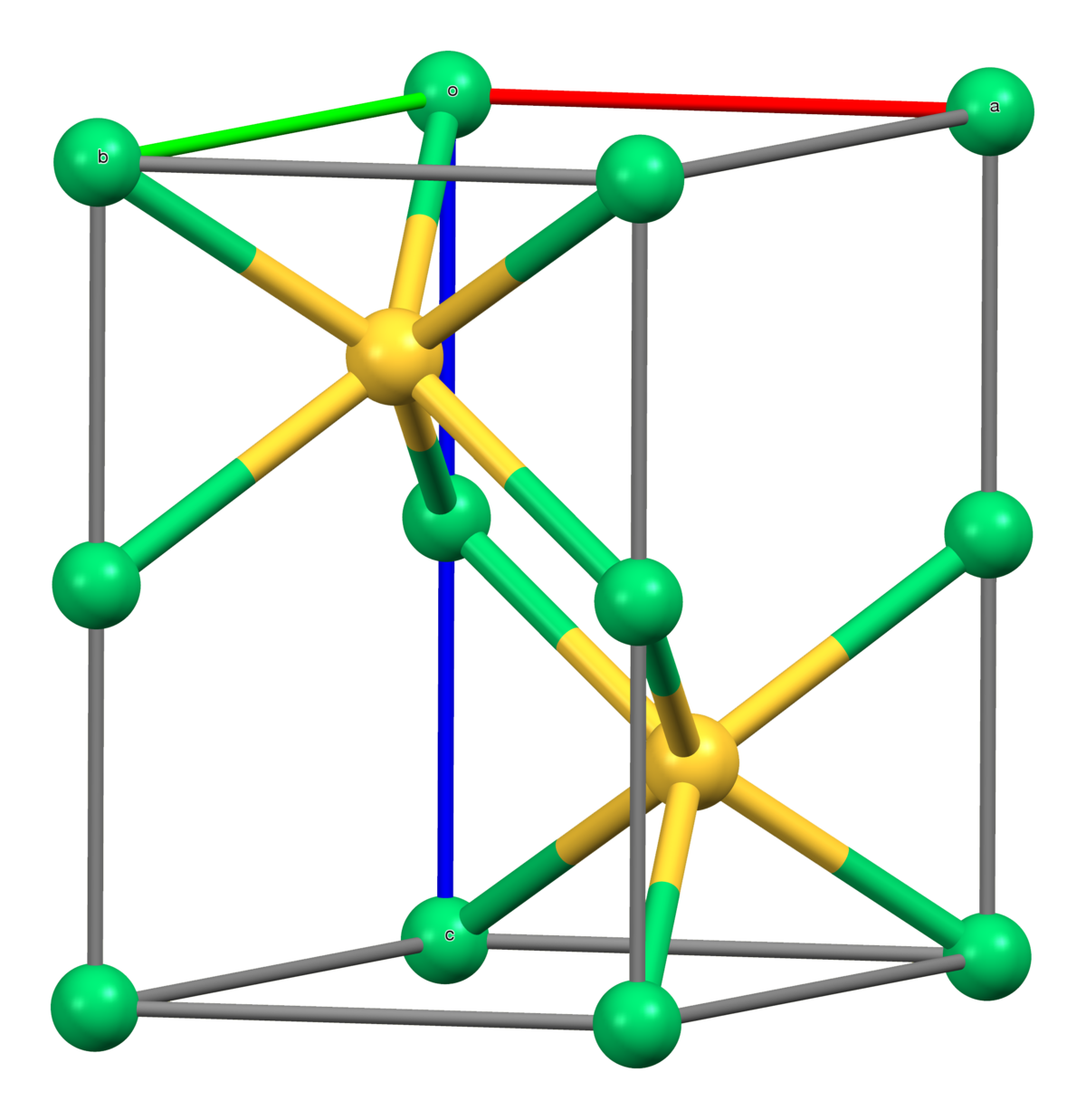



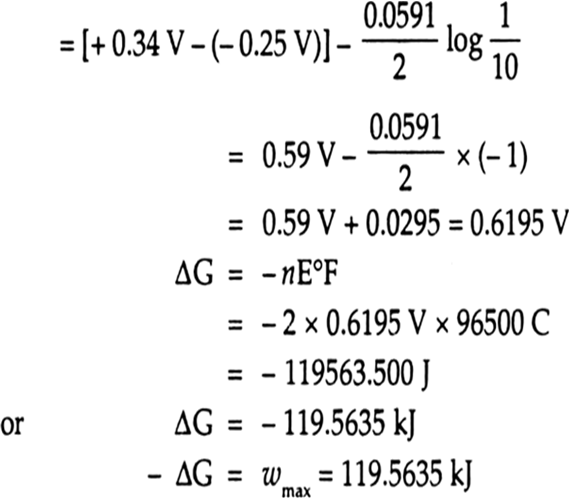
![PDF] Raman Spectroscopy of Nickel Sulfide Ni | Semantic Scholar PDF] Raman Spectroscopy of Nickel Sulfide Ni | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/345ef7d84582812e979cbd9a8f6545615d4ef1a4/2-Figure4-1.png)